This story additionally ran on NPR. This story might be republished free of charge (details).
David Herzberg was alarmed when he heard that Richard Sackler, former chairman of opioid big Purdue Pharma, was listed as an inventor on a brand new patent for an opioid dependancy remedy.
Patent No. 9861628 is for a fast-dissolving wafer containing buprenorphine, a generic drug that has been round since the 1970s. Herzberg, a historian who focuses on the opioid epidemic and the historical past of pharmaceuticals, mentioned he fears the patent may maintain costs excessive and make it harder for poor addicts to get remedy.
“It’s hard not to have that reaction of, like … these vultures,” mentioned Herzberg, an affiliate professor on the University of Buffalo.
James Doyle, vice chairman and basic counsel of Rhodes Pharmaceuticals, the Purdue subsidiary that holds the patent, mentioned in an electronic mail assertion that the corporate doesn’t have a developed or accepted product and “therefore no money has been made from this technology.”
“The invention behind the buprenorphine patent in question was developed more than a dozen years ago,” he wrote. “If a product is developed under this patent, it will not be commercialized for profit.”
Yet, the patenting of a small change in how an current drug is made or taken by sufferers is a part of a tried-and-true pharmaceutical trade technique of enveloping merchandise with a sequence of protecting patents.
Drug corporations sometimes have lower than 10 years of unique rights as soon as a drug hits the marketplace. They can prolong their monopolies by layering in secondary patents, utilizing ways critics name “evergreening” or “product-hopping.”
Lisa Larrimore Ouellette, a patent legislation skilled at Stanford University, mentioned the pharmaceutical trade will get a better monetary return from its patent technique than that of some other trade.
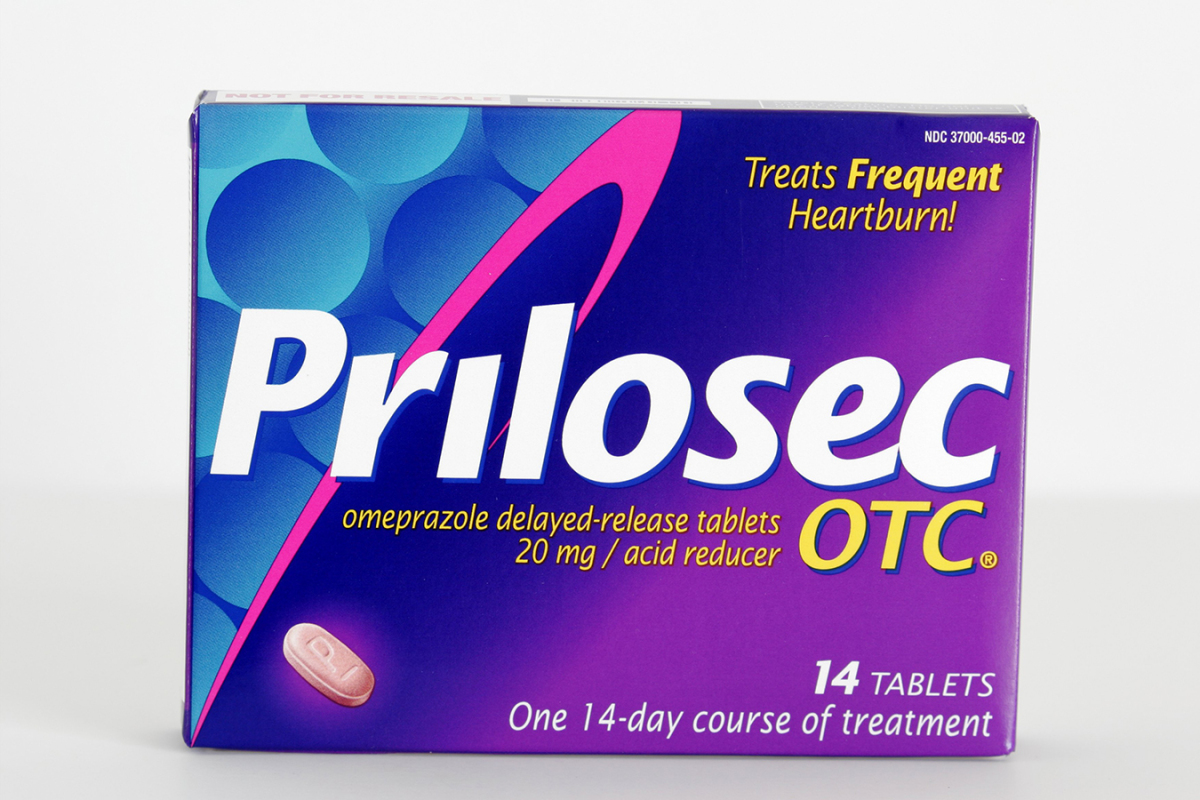
AztraZeneca in 2001 famously fended off generic variations of its blockbuster heartburn medication Prilosec by patenting a tweaked model of the drug and calling it Nexium. When Abbott Laboratories confronted a number of generic lawsuits over its massive moneymaker Tricor, a decades-old ldl cholesterol drug, it lowered the dosage and altered it from a pill to a capsule to win a brand new patent.
And Forest Laboratories stopped promoting its Alzheimer’s illness drug Namenda in 2014 after reformulating and patenting Namenda XR to be taken as soon as a day as an alternative of twice.
Another widespread technique is to create what Food and Drug Administration Commissioner Scott Gottlieb calls “patent thickets,” claiming a number of patents for a single drug to construct safety from opponents. AbbVie’s rheumatoid arthritis drug Humira has gained more than 100 patents, for instance.
Email Sign-Up
Subscribe to KHN’s free Morning Briefing.
The U.S. Patent and Trademark Office awards patents when an innovation meets the minimal threshold of being new and non-obvious. Secondary patents are routinely granted to established medicine when an enchancment is made, akin to making it a once-a-day capsule as an alternative of twice a day, mentioned Kristina Acri, an economist and worldwide mental property skilled on the Fraser Institute and Colorado College.
“Is there a better way? Maybe, but that’s not what we’re doing,” Acri mentioned.
The controversial patent that Sackler and 5 co-inventors obtained is broadly often known as a “continuation patent.” (The unique patent utility for the wafer was filed in August 2007.)
Continuation patents don’t essentially prolong the patent lifetime of a drug, however they will produce other makes use of. In 2016, Rhodes filed a lawsuit towards Indivior alleging patent infringement.
Indivior, previously a part of Reckitt Benckiser, sells a movie model of the favored dependancy remedy drug Suboxone that’s positioned below the tongue — an oral medication just like what Rhodes has patented. Indivior’s is available in a lime taste.
Indivior’s movie, which federal regulators accepted in 2010, dominates the market with a 54 p.c common market share, in accordance with the corporate’s most up-to-date monetary report. And the corporate has vigorously fought rivals, together with submitting lawsuits towards companies akin to Teva Pharmaceutical Industries, which sought approval to fabricate generic variations. Indivior declined to remark.
The Rhodes Pharmaceuticals model could be a wafer that melts shortly within the mouth. The inventors checklist potential flavors together with mint, raspberry, licorice, orange and caramel, in accordance with the patent.
For opioid historian Herzberg, the patent battles between corporations like Rhodes and Indivior are “absolute madness.”
Decisions on what is out there in the marketplace to deal with addicts ought to be based mostly on what’s the easiest way to deal with the individuals who have the issue, he mentioned.
Patent battles, Herzberg mentioned, are “not how you want drug policy getting made.”
Attempts to vary the patent system have intensified over the previous decade as costs of pharmaceuticals continue to climb.
In 2011, President Barack Obama signed the America Invents Act, which included the creation of the Patent Trial and Appeal Board. The PTAB is a substitute for utilizing the cumbersome U.S. court docket system to problem weak patents. Generic drug producers have used the board’s “inter partes review” course of and overturned 43 p.c of the patents they challenged, in accordance with recent research.
Critics of the executive course of, together with the pharmaceutical trade commerce group PhRMA, mentioned it creates “significant business uncertainty for biopharmaceutical companies.” Often corporations should defend their merchandise twice — each within the courts in addition to earlier than the PTAB, mentioned Nicole Longo, PhRMA’s director of public affairs.
Drug big Allergan attempted to beat the PTAB’s evaluate course of by arguing that the patent couldn’t be challenged on the evaluate board as a result of they bought the patent to the St. Regis Mohawk Tribe, which had sovereign immunity. A federal appeals court docket ruled this summer that Allergan couldn’t defend its patents from the PTAB evaluate this manner.
This 12 months, a number of members of Congress proposed payments that will unwind or restrict modifications made by the America Invents Act, although nothing is more likely to occur earlier than the midterm elections. The STRONGER Patents Act, launched in each the House and Senate, would weaken the PTAB board by aligning its claims requirements with what has been established by court docket rulings.
This story additionally ran on NPR. This story might be republished free of charge (details).
KHN’s protection of prescription drug improvement, prices and pricing is supported partially by the Laura and John Arnold Foundation.
Sarah Jane Tribble: [email protected]”>[email protected], @SJTribble
Related Topics Health Industry Pharmaceuticals Drug Costs FDA Opioids Prescription Drugs src=”http://platform.twitter.com/widgets.js” charset=”utf-Eight”>