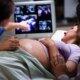
Lorraine Bonner, a retired Oakland, Calif., physician who’s now a sculptor, says she spent a 12 months recovering after surgical staples have been used to seal her colon. (Heidi de Marco/KHN)
Lorraine Bonner felt as if she was the one one. The surgical staples used to seal her colon after surgical procedure had leaked, she has alleged in a lawsuit, spurring further surgical procedures and an extended, troublesome restoration.
And then, simply over a 12 months after the ordeal, she learn a Kaiser Health News investigation that described worse circumstances. KHN revealed that the Food and Drug Administration had allowed stapler maker Covidien to quietly file tens of 1000’s of studies of stapler malfunctions right into a then-hidden database.
Alarmed that others had been harmed and studies had been hidden, Bonner, a retired Oakland, Calif., physician, determined to go ahead with a lawsuit in opposition to the stapler maker.
“If the information had been out there, then maybe Covidien would have changed the design of the staplers and made them safer,” she stated, “and that would have obviated the problem in the first place.”
Bonner’s lawsuit is one instance of how an unlimited cache of data that have been launched this summer season are taking up a lifetime of their very own.
For virtually 20 years, malfunctions and accidents linked to 108 medical gadgets, together with dental implants and pacemaker leads, have been funneled into an FDA database that few sufferers, docs and even FDA officers knew existed.
In 2016, for instance, Covidien reported 84 accidents or malfunctions within the public database generally known as MAUDE, whereas almost 10,000 incidents flowed into the hidden database, KHN reported in March. A couple of MAUDE studies talked about the existence of an “alternative summary reporting” program, however till this summer season, the FDA made that inside knowledge out there solely by the Freedom of Information course of, which might take as much as two years.
KHN’s investigation prompted then-FDA commissioner Scott Gottlieb to pledge in a tweet to open the hidden knowledge to the general public. The company launched all 5.7 million data in June.
Since then, researchers, attorneys and the FDA’s personal officers have taken a more in-depth take a look at the information to be taught extra about which gadgets malfunctioned, and the way usually.
Libbe Englander, the founder and CEO of Pharm3r, a medical knowledge consulting agency, found that the gadgets within the hidden database have been a lot riskier than different gadgets tracked by the FDA.
Her agency concluded that the hidden studies have been “more likely to be associated with life-threatening devices and to contain potentially serious problems.”
For instance, simply 10% of the gadgets tracked within the MAUDE database are implanted within the physique. But 44% of these within the hidden knowledge are lodged in a affected person’s physique, together with pacemakers and coronary heart valves.
The Pharm3r report additionally discovered that the gadgets within the hidden knowledge have been extra prone to be topic to a Class 1 recall, initiated when a tool downside may trigger severe damage or loss of life. The report additionally underscored how huge the now-open knowledge is ― accounting for about 40% of the entire device-problem studies lodged with the FDA over the previous 20 years.
The once-hidden studies additionally figured into the ECRI Institute’s annual checklist of well being expertise hazards, a listing circulated to hospitals and well being techniques.
ECRI discovered that this 12 months’s No. 1 hazard was misuse of the surgical stapler ― which has been linked to 412 deaths, greater than 11,000 severe accidents and almost 100,000 malfunctions since 2011, based on the FDA.
“Most of these [stapler] reports had not previously been accessible to the public,” the ECRI report notes. ECRI has suggested docs to have a backup plan in place in case a stapler malfunctions throughout surgical procedure.
“If a hazard is getting a lot of attention, hospitals want to have a plan to address that,” stated Rob Schluth, a senior venture officer with the institute.
Bonner says her lengthy, troublesome restoration after a number of surgical procedures stored her from tending to her backyard for a few 12 months. (Heidi de Marco/KHN)
For Bonner, the issues with a surgical stapler grew to become obvious simply over every week after she had colon surgical procedure in November 2017. Retired from a profession as a hospital doctor, she knew the steps to take towards restoration. But she felt nauseated on a regular basis and will barely maintain down water.
“I was trying to get up and walk,” she stated. “I was doing everything, but I just got worse and worse.”
Doctors went in for a second surgical procedure on Dec. 6 and located contaminated pus filling her stomach. They routed her colon’s contents to be collected outdoors her physique. It took further surgical procedures to revive her colon operate and tackle hernias brought on by the procedures.
Bonner’s lawsuit alleges that a Covidien stapler prompted her bowel leak and that the corporate, by not reporting malfunctions to the general public database for years, “have hidden the true risks of using the device from surgeons and patients.”
In court docket data filed within the ongoing case, Covidien denied all of Bonner’s claims however did say that the FDA invited the corporate in 2001 to submit stapler malfunctions with quarterly “alternative summary” studies.
Medtronic spokesman John Jordan stated the corporate doesn’t have a touch upon the pending authorized case.
Other gadgets are underneath scrutiny. The once-hidden knowledge showed more than 2 million issues with dental implants.
During an October FDA webinar on dental gadgets, an FDA official stated her workplace was “taking a deeper dive and trying to identify lessons learned” from dental implant incidents.
Dr. Malvina Eydelman, of the Division of Dental Devices within the company’s gadget department, acknowledged that the variety of occasions was “very high” and stated her workforce was making an attempt to develop a plan to share info with the general public.
Also underneath scrutiny are the textured breast implants made by Allergan. The firm’s Biocell implants, recalled in July, have been linked to 12 deaths and 481 of the 573 worldwide circumstances of “breast implant-associated” lymphoma, based on the FDA. Breast implant accidents and malfunctions accounted for almost half 1,000,000 studies within the hidden database, together with implants that leaked, deflated or migrated.
Patients in Missouri, California, New York and Illinois have filed federal class motion lawsuits in opposition to Allergan in current weeks, every decrying the corporate’s heavy use of the “alternative summary reporting” program to file damage studies ― which have been analyzed by the International Consortium of Investigative Journalists.
Bonner’s lawsuit alleges that a Covidien stapler prompted her bowel leak. Bonner says that Covidien hid “the true risks of using the device from surgeons and patients.” (Heidi de Marco/KHN) Bonner determined to go ahead with a lawsuit in opposition to stapler maker Covidien after studying a KHN investigation describing worse circumstances of stapler malfunctions. (Heidi de Marco/KHN) Bonner, a retired physician who’s now a sculptor, works in her dwelling studio in Oakland. (Heidi de Marco/KHN)
A category motion case filed in Florida notes FDA guidelines say that different abstract studies can not embody extreme or sudden occasions, “yet it is believed that these incidents were kept hidden in ASRs,” together with a minimum of one lymphoma case.
An ongoing federal securities lawsuit additionally alleges that Allergan used “inappropriately nonpublic” different abstract studies, disregarding the “risk that some [lymphoma] cases would go undetected by the FDA.”
Allergan has not but responded to the category motion circumstances or to requires remark. It denied the “threadbare” allegation within the securities lawsuit, citing the KHN article, which “makes clear that the filing of ASRs was permissible and even encouraged by the FDA to reduce ‘redundant paperwork.’”
While the FDA has ended the choice abstract reporting program, it opened the door for makers of greater than 5,000 gadget varieties to file quarterly summaries of reported malfunctions. In every case, the gadget maker should file a public report back to the MAUDE database noting what number of studies have been filed straight with the company.
An FDA spokeswoman stated the company has stopped accepting abstract studies of accidents cited in pelvic mesh lawsuits. The company does proceed to just accept a whole lot of loss of life report summaries underneath a particular exemption for gadgets tracked in a “registry” held by a specialty medical society.
“We are in the process of reviewing the effects of these changes and will continue working to improve the usability and transparency of information in the MAUDE database — whether that information was submitted as a summary or individual report,” FDA spokeswoman Stephanie Caccomo stated in an e-mail.
The launch has not been as informative as Madris Tomes had hoped, she stated, provided that the FDA didn’t disclose knowledge on how the gadgets harmed sufferers. Tomes is a former FDA supervisor who runs the corporate Device Events, which analyzes FDA knowledge.
For occasion, she stated a breast implant damage report would possibly say there was a “biocompatibility” downside, nevertheless it’s unclear what occurred to the affected person.
“Did it result in lymphoma or the patient needing a device replacement?” requested Tomes. “Without that outcome, we’re still a little bit in the dark on what this data means.”