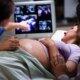
Rachel Glenn likes to puree meals and put it in her Four-year-old’s feeding tube. The Arkansas mother mentioned her thick concoctions of fruit, greens, meat and grains present the concentrated vitamin he wants.
When he was on components, Asher, who can’t swallow attributable to a brainstem situation, vomited a number of instances a day. Since his mother began making his meals in a blender, when he was about 15 months outdated, that has stopped and Asher has extra vitality. Plus: “He gets his two cups of greens a day and he doesn’t argue about it.”
So Glenn was upset to find out about a brand new kind of connector being promoted for feeding-tube gear that might forestall her son from consuming her home-blended meals. It has a one-size-fits-all opening that’s a lot narrower than the holes on syringes she presently makes use of to push meals by means of the feeding tube implanted in his stomach.
Since Rachel Glenn began making meals in a blender for Four-year-old son Asher, when he was about 15 months outdated, he has stopped vomiting all through the day and has extra vitality. Asher can’t swallow attributable to a brainstem situation. (Courtesy of Rachel Glenn)
When Glenn and her husband tried utilizing a syringe tipped with one of many new plastic connectors, referred to as ENFit, they might barely get Asher’s meals to budge. She worries her son’s well being will undergo if producers begin placing ENFit connectors on all feeding gadgets.
“We feel being forced back onto formula is unfair to him,” mentioned Glenn. “I don’t see any evidence that they have taken the needs of actual tube-feeding families into consideration.”
As the brand new connector commonplace meant to forestall lethal errors in hospitals has been launched within the area of interest machine trade, sufferers and their households who use feeding tubes long run at dwelling say their issues have been largely ignored. The capacity to eat regular meals and management their vitamin is a valuable profit they worry might vanish.
Some critics say ENFit additionally creates new dangers, together with dosing errors and infections amongst preemie infants.
ENFit sprung from an trade effort backed by affected person security teams and the Food and Drug Administration to create a world system of connectors that might forestall gadgets with totally different features from being linked.
“We feel being forced back onto formula is unfair to him,” says Glenn of her younger son. “I don’t see any evidence that they have taken the needs of actual tube-feeding families into consideration.” (Courtesy of Rachel Glenn)
Both the Joint Commission, which accredits hospitals, and the World Health Organization flagged such misconnections as a threat greater than a decade in the past. In 2010, The New York Times drew attention to the issue with a case of a pregnant lady who died after a bag of components was related to her IV port.
An trade committee, led by an FDA official, convened to set a regular for feeding-tube connectors. Some producers started investing in manufacturing and advertising and marketing years earlier than the usual was permitted in 2016.
At some level, “the ‘P’ that stood for patient safety that should have been in the center of decision-making was changed for a ‘P’ that stood for profit,” mentioned Maureen Burger, chief nursing officer at Visante, a pharmacy consulting agency based mostly in St. Paul, Minn. Her agency has served as a guide for Becton Dickinson and Co., a number one syringe maker.
Unintended Consequences
ENFit syringes have an empty area on the tip the place medicine and meals can get trapped, doubtlessly resulting in bacterial contamination in addition to dosing inaccuracy that may be particularly hazardous for tiny infants. One study discovered ENFit syringes delivered the unsuitable dose a few fifth of the time.
Babies with neonatal abstinence syndrome are weaned off medication — usually with morphine each three hours for 30 days, mentioned Keliana O’Mara, a pharmacist on the University of Florida Health Shands Hospital in Gainesville, Fla., who co-authored the research. “That’s 240 chances to have a problem with dosing accuracy.”
That hasn’t stopped a commerce group from pushing hospitals to transform all their gear — together with the feeding tubes implanted in sufferers in addition to tubes and syringes used to ship meals and medication — to ENFit. In July, the Global Enteral Device Supplier Association startled many individuals by announcing that its member producers would start phasing out non-ENFit merchandise subsequent 12 months.
In response to questions, affiliation Executive Director Michael Cusack referred to a document on the group’s “Stay Connected” web site that claims there have been “no reported misconnections, dosing concerns, or major transition problems” with ENFit within the United Kingdom, the place adoption has been widespread.
Email Sign-Up
Subscribe to KHN’s free Morning Briefing.
While the doorway of recent connectors is inflicting misery for dwelling customers — who name themselves “tubies” — some specialists say it has not achieved a lot of something to handle the issue it was purported to resolve.
“It was poorly designed, it was poorly thought out, and it doesn’t get at the root of the misconnections,” mentioned Shelley Buma, who owns a medical machine design agency in Massachusetts.
While the suppliers affiliation highlights tragic instances of sufferers damage or killed by misconnections, a few of its critics say ENFit doesn’t repair any misconnection hazards. They say it could not have prevented any of 23 feeding-tube misconnection occasions reported to the FDA between 2005 and 2016.
“That’s really a point of outrage for me,” Buma mentioned.
By the time the design was permitted, misconnection dangers with feeding tubes had largely been mitigated, at the least within the U.S., mentioned Buma.
What’s extra, she added, ENFit’s design created pointless challenges for the rising inhabitants of tubies, a lot of whom use funnel connectors and catheter tip syringes with huge openings that she mentioned don’t have any threat of misconnection with different gadgets.
Beyond issues with blended meals, tubies complain of slower circulate charges for components, the clogging of medicine and a screw-on design that’s a barrier for folks with restricted use of their fingers, similar to these with arthritis.
The Push To Switch
The suppliers affiliation has petitioned for the FDA, the Centers for Medicare & Medicaid Services and the Joint Commission to take steps that might compel common adoption. So far, that hasn’t occurred.
While there’s no mandate, some hospitals got here to consider a shift to ENFit was inevitable. “The message most people got was, ‘You have to convert because it’s going to be required in the future,’” mentioned O’Mara, whose personal hospital system aborted a change to ENFit final 12 months after taking a better take a look at the impact on affected person care.
And reluctance to make the change seems to be widespread. A suppliers affiliation survey in 2018 confirmed 28% of U.S. hospitals had transitioned to ENFit, although some trade specialists mentioned they consider the adoption price is decrease.
Some producers even have resisted a change to ENFit. Becton Dickinson left the provider affiliation in 2015 citing ENFit dosing security. The firm mentioned it’s growing an alternate design that will probably be obtainable to all producers and can adjust to the usual and not using a misdosing threat.
French neonatal producer and affiliation member Vygon and Ohio-based feeding-tube maker Applied Medical Technology have mentioned they are going to hold non-ENFit gadgets in the marketplace.
Still, the push for ENFit has left some tubies feeling burned. They are offended that components makers similar to Nestle and Abbott Laboratories served on the requirements committee and joined the suppliers affiliation, although it’s unclear whether or not these corporations held sway in choosing a design that finally seems to serve their pursuits.
Nestle and Abbott declined to touch upon their involvement.
Tubies fear their trusty gadgets might disappear, forcing them to depend on industrial components that’s costlier and, some consider, much less nutritious than common meals.
“It’s my great concern that as more and more unknowing tubies are supplied with ENFit, the demand for legacy products will be diminished,” mentioned David Rowland, an 80-year-old South Carolina resident who co-founded a bunch referred to as Tubies Against ENFit.
Some, just like the Glenn household, say they’re hoarding provides, simply in case.
Related Topics Cost and Quality Health Industry Children’s Health Chronic Disease Care FDA Nutrition